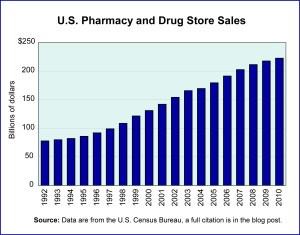
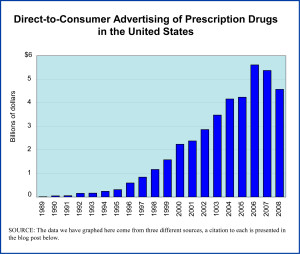
Year: 2020 and 2028
Market size: $7.21 billion and $13.38 billion, respectively
AARP’s 2019 Brain Health and Dietary Supplements Survey found that more than 25% of Americans aged 50 and over regularly take brain health supplements. As the population ages, the incidence of Alzheimer’s disease and other forms of dementia increases. As of 2020, about 5.8 million people in the United States had Alzheimer’s; 50 million worldwide had some form of dementia.1 The World Health Organization (WHO) estimates that there are 10 million new cases of dementia yearly — one case diagnosed every 3 seconds worldwide. The fear of loss of brain function due to aging is contributing to the adoption of brain health supplements among older adults.
The increasing awareness of mental health issues among adolescents and millennials is fueling the demand for brain health supplements among these age groups. These consumers seek treatment for depression, stress, and anxiety, all common conditions among younger people. The COVID-19 pandemic increased the occurrence of these conditions and in turn increased the demand for brain health supplements to treat them.
Overall, by application, memory enhancement garnered the largest revenue share in 2020, more than 25%. This was followed by attention and focus, depression and mood, sleep and recovery, anti-aging and longevity, and stress and anxiety. The depression and mood segment is expected to see the fastest growth through 2028 due to the increasing rate of depression among millennials and the growing awareness of mental health and depression in countries such as India and Indonesia. According to the WHO, more than 264 million people worldwide suffer from depression.
Natural molecules accounted for more than half the market in 2020, likely due to their cost-effectiveness and ready availability. Omega-3 fatty acids, Huperzine-A and Acetyl-L-carnitine, for example, are used to improve focus and to treat depression, stress, and anxiety. Docosahexaenoic acid (DHA), eicosapentaenoic (EPA), and Alpha-linolenic acid (ALA) are three major omega-3 fatty acids used to treat depression and cognitive impairment, including Alzheimer’s disease.
Revenues for herbal extract products are expected to grow at the fastest rate through 2028 as interest in herbal supplements rises in such countries as the United Kingdom and the United States. Demand for ginkgo biloba, ginseng, lion’s mane, curcumin, echinacea, and bacopa monieri has been increasing in recent years. The vitamins and minerals segment has the smallest market share.
A large number of global and regional manufacturers have a presence in the industry. These companies have been using a combination of new product launches, innovation, mergers and acquisitions, and capacity expansion to increase their shares of the market. Some of the larger manufacturers include Teva Pharmaceutical Industries Ltd., HVMN Inc., Purelife Bioscience Co. Ltd., Peak Nootropics, AlternaScript, Liquid Health Inc., Natural Factors Nutritional Products Ltd., Reckitt Benckiser Group PLC, Onnit Labs Inc., and Quincy Bioscience. Leading brands include The Genius Brand, Bulletproof, Onnit Labs, Vital Vitamins, Prevagen, and Neuriva. Of the 348 brands sold on Amazon, only 18 brands garnered above a 1% share in 2020. The top 18 brands represented 56% of the market, with the other 330 accounting for the remaining 44%.
1 Figure for the United States is from the Alzheimer’s Association. Worldwide figure is from the World Health Organization.Sources: “Global Brain Health Supplements Market Size, Share & Trends Analysis Report by Product (Natural Molecules, Herbal Extract), by Application (Memory Enhancement, Depression & Mood), by Region, and Segment Forecasts, 2021-2028,” Research and Markets Report Description, January 2021 available online here; “Brain Health Supplements Market Size, Share & Trends Analysis Report by Product (Natural Molecules, Herbal Extract), by Application (Memory Enhancement, Depression & Mood), by Region, and Segment Forecasts, 2021 – 2028,” Grand View Research Report Overview, January 2021 available online here; “Global Brain Health Supplements Market Size, Share & Trends Analysis Report 2021 – ResearchAndMarkets.com,” Business Wire, September 9, 2021 available online here; Sinan Atac, “Nootropic Supplement Sales on Amazon,” Natural Products Insider, April 2021, pp. 39-40 available online here.
Image source: formulateHealth, “Health_Supplements_-_Nutraceuticals_-_50191152323.jpg,” Wikimedia Commons, CC BY 2.0, July 30, 2020 available online here.